The Biochemical Sequence
© 2014 by Hugh Lovel
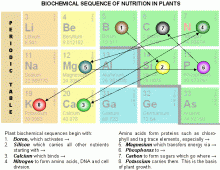
What is the hierarchy or ‘biochemical sequence’ of what must function first before the next thing and the next thing works. The elements early in this sequence must be present and working well before later elements have any chance of being useful for plant growth. Nitrogen, phosphorous and potassium occur late in this biochemical sequence, while sulphur, boron, silicon and calcium start things off.
0 Sulphur: Sulphur interacts with life chemistry (carbon-hydrogen-oxygen-nitrogen compounds) at surfaces. Along with warmth, it is the principle catalyst in biochemistry. Since everything going on in the soil biology occurs at the surfaces of soil particles where minerals react with water, air and warmth, sulphur activates surfaces—is the essential ‘key-in-the-ignition’ for kicking off robust soil biochemistry. In his Agriculture Course, Steiner speaks of how ‘the spirit-activity of the universe works as a sculptor, moistening its fingers with sulphur . . .’ [1]
Along with warmth, it is the classic catalyst of carbon chemistry.
Sulphur works at the surfaces, boundaries and edges of things to bring organization and life into being. Regardless of other soluble elements, the soluble soil test for sulphur should show 50 ppm sulphur [Morgan test] for biological soil fertility to function properly. Light soils may need a bit less and heavy soils may need more. In the total test a 60 to 1 carbon to sulphur ratio is helpful to ensure enough sulphur in soil reserves.
Silicon forms the basis for the capillary action that takes up nutrients from the soil. Fortunately for agriculture, silicon’s activity defies gravity. But to do this silica relies on boron, a component of clay. In his second agricultural lecture Steiner insightfully asserts, “First we need to know what is really going on. However else clay may be described, however else we must treat it so that it becomes fertile—all this is of secondary importance; the primary thing we need to know is that clay promotes the upward stream of the cosmic factor.”[2]
1 Boron: It is the boron component in clay that is the accelerator pedal of agriculture, while silicon forms the highway that carries nutrients throughout plants and animals. Boron interacts with silica in the linings of transport vessels and stimulates the flow of nutrients along the silicon highway. This places boron first in the biochemical sequence, and if either boron or silicon is deficient the soil biology will function below its potential. With either boron or silicon deficiency—and especially with both—crops will wilt instead of growing on hot days. Ironically, the two most effective ways to create boron and silicon deficiency are:
1. Clean cultivation
2. Use of artificial nitrogen fertilisers
Though standard in modern agriculture, these practices make boron and silica available by killing off the soil biology that builds and maintains the soil’s clay/humus complexes. This releases a flush of boron and silicon which can easily drain way through the landscape.
2 Silicon: Of course, sap pressure would be no use without a transport system to contain it, and silicon provides the actual transport of nutrients. Interestingly, applying too much boron too early in a crop cycle is notable for burning seedlings and young transplants-such as sprouting squash, beans or tomatoes-because too much sap pressure in such a tiny plant drives sodium out the leaf margins. Nevertheless, in plants where leaf veins are highly branched, like peas, beans, squash and tomatoes, boron is important in later growth to maintain strong enough sap pressure to make such a complex system work.
On the other hand, highly siliceous plants, such as grasses, do well on less boron to give them sap pressure since their transport vessels all run parallel without branching. That’s like irrigation lines that only feed one sprinkler head. Such a thing doesn’t take much pressure.
Obviously without robust transport, nowhere near as much nutrient reaches the leaves or is stored in the fruits. Chemical agriculture gets around this to some extent, since-even with a weak transport system-anything that is highly soluble, such as potassium nitrate, is simply taken up along with water. Though this dilutes the sap, it flows quite easily due to low sap density. This is why chemically grown foods commonly have coarse, watery cell structure, as well as lower nutrition and poorer keeping quality. However, without a robust transport system, heavier, less-soluble nutrients such as calcium, magnesium, carbohydrate-and-amino-acid complexes can easily be left behind.
3 Calcium, which comes next in the sequence, is the truck that travels on the highway. Along with magnesium, potassium and sodium calcium forms the lime complex traffic that dominates the reactive side of life chemistry.
Where silicon, along with carbon forms the weakly-reactive nutrient highway, calcium, along with oxygen, forms the strongly reactive cargo that flows down the silica transport and containment system. Calcium and the lime complex is the last thing you want to leave behind because of its role in nitrogen fixation and amino acid chemistry. Calcium balances charge in proteins and is particularly important in cell division, which is the first thing that happens in fruit or seed formation after pollination. Without it there would be no fruit or seed. It collects and carries with it the nutrients that follow in the biochemical sequence.
As the opposite polarity of plant chemistry from the free-handed silicon, calcium is hungry, even greedy. This is why it needs the aloof silica to line the transport system. Above all else, calcium engages nitrogen to make amino acids, the basis of DNA, RNA and proteins. In turn, these nitrogen compounds are responsible for the complex enzyme and hormone chemistry of life which employs everything from sulphur and silicon to magnesium, iron, phosphorous, zinc, manganese copper and other trace elements. Probably the most important point is, nitrogen provides the amino acids in chlorophyll, which is key to photosynthesis, a highly efficient means of catching energy.
For example, taking corn, Zea maize, if calcium does not reach the ear in sufficient quantities, the kernels near the end of the ear simply do not fill out. With a crop like soybeans Glycene max, double or even triple the calcium values of maize are needed for full pod set without shedding pods-a common problem in soybeans. Wouldn’t you like to see every kernel on your maize fill out to the end of the ear and every soybean blossom produce a full pod of beans? This only happens when boron, silicon and the calcium lime complex work together well.
4 Nitrogen: As just mentioned, wherever calcium goes there also goes nitrogen. And nitrogen is the basis of amino acid formation, protein chemistry and DNA replication and expression. Once nitrogen enters the picture all sorts of proteins, enzymes and hormones are produced and very complex things are set in motion involving trace elements.
Unfortunately, soluble nitrogen fertilisers only stimulate this latter portion of the sequence without addressing the priorities of sulphur, boron, silicon and calcium. Such fertilisers stimulate growth, but they are like methamphetamine. They grow weak crops that depend on growing in weedy conditions where they fall prey to pests and diseases.
All parts of a plant’s protein chemistry require amino acid nitrogen. Nitrogen straddles the divide between the chemically indifferent silicon and the calcium large amounts of amino acids go into the formation of chlorophyll where energy is gathered. After all, gathering and sequestering energy is essential to life. Without photosynthesis plants would never grow. This is where magnesium, phosphorous, potassium and a wide range of micronutrients follow nitrogen in the biochemical sequence.
5 Magnesium: Since photosynthesis requires magnesium, it is fifth in the biochemical sequence, ahead of all the more minor trace elements.
Of course, photosynthesis is not simply a matter of chlorophyll catching energy. The energy has to be transferred from the chlorophyll to the silicon into producing sugars out of carbon dioxide and water, which requires phosphorous for energy transfer. Otherwise the chlorophyll burns up, and the leaves turn a wine red colour.
However, as long as there is enough phosphorous, carbon is pried loose from carbon dioxide so it can combine with water to make sugar and release oxygen.
6 Phosphorous: Of course, photosynthesis is not simply a matter of chlorophyll catching energy. The energy has to be transferred into producing sugars out of carbon dioxide and water, which requires phosphorous for energy transfer. Otherwise the chlorophyll burns up, and the leaves turn a wine red colour.
7 Carbon: As long as there is enough phosphorous, carbon is engaged as carbon dioxide and the energy transferred from chlorophyll via phosphorous to combines carbon dioxide with water, making sugar and releasing oxygen.
8 Potassium: At this point the sugars pass into the plant’s sap where potassium, the electrolyte, guides them to wherever they most need to go.
Yes, Oversimplified
Understandably, this sequence is oversimplified. For example, sulphur is the classic catalyst in carbon (organic) chemistry. Without it, nothing-not even the boron-would give rise to life. Also, potassium has a very close relationship with silicon, so when silicon carries calcium and amino acids to the cell division sites in the plant, potassium plays the role of an electronic doorway that lets the calcium and amino acids enter the cells that are preparing to divide. If cold weather slows potassium down, or if it is in short supply, then calcium and amino acids cannot reach the cell nuclei, the DNA cannot divide, cell division fails and the fruit falls off the plant. Sometimes entire fruit crops are lost to a couple degrees of frost when a light spray of kelp with potassium silicate would save the day.
Supplementation with Minerals and Rock Powders
Even though quantum agriculture is primarily about organization and biological activities, soil mineralization must be considered. How does one organise something if it isn’t there? Many soils need gypsum or elemental sulphur because they are sulphur deficient in both their soluble and total tests. Many soils also need silicon rock powders—also a source of boron. This is true if past nitrogen fertilisation has flushed whatever boron and silicon was there away. Boron and silicon deficiencies also occur following overgrazing or clean cultivation. Silicon availability may need to be fostered to get the soil biology up and running so it can release more silicon from the surfaces of soil particles. The soil’s silicon biology is easily depleted by nitrogen fertilization, overgrazing or clean cultivation.
Through lack of experience and understanding, many ‘organic’ farms use raw manures—the worst being chicken manure—as a nitrogen source. This soon depletes sulphur, boron and silicon. The remedy for this is likely to be compost made by adding 10% or so of high silicon rock powders along with a little gypsum to composts and composting fully with soil until it looks and smells like soil.
In addition to gypsum and high silica rock powders, lime can be used to provide calcium. Dolomite also provides magnesium if this is needed. Rock phosphorous provides silicon, calcium and phosphorus. There are also natural potassium sulphate ores. Rock powders tend to also provide a variety of trace elements. For high pH soils with large excesses of sodium and potassium the remedy in drier climates may be increasing the soil’s holding capacity with humates and zeolite to buffer pH and build more storage.
What’s the Aim?
Most importantly, the biochemical sequence shows us we need to start with sulphur to expose the surfaces of soil particles to biological activity so reserves can kick in. Other methods may not recognize sulphur’s key importance, but in quantum agriculture this should be clear. And where budgets are slim and long range soil fertility is desired boron, silicon and calcium follow sulphur in importance.
Unfortunately for nutrition, health and long term vitality of the soil’s biochemistry, soluble NPK fertilisers continue to be used for their ability to gloss over deficiencies of sulphur, boron, silicon and calcium. Large reserves of nitrogen, as well as phosphorous and potassium, are commonly present—even if inactive—at the surfaces of soil particles where the organization of life chemistry arises. Only when the biochemistry of sulphur, boron, silicon and calcium is thriving can the potential of these reserves become available.
This all goes back to Liebig’s ‘law of the minimum’ which says plants can only perform as well as their most deficient nutrients.
[1] Agriculture, Rudolf Steiner, Creeger-Gardner translation, pp 44-47.
[2] Agriculture, Rudolf Steiner, Creeger-Gardner translation, page 31.